Introduction
|
X-linked myotubular myopathy is a recessive, sex linked disorder that results in severe and debilitating disease often resulting in death within the first few years of life. Genetically, XLMTM can lead to a heterogeneous group of congenital conditions seen at birth including muscle weakness (myopathy) and decreased muscle tone (hypotonia) affecting muscles through the body. Decreased tone in skeletal muscle is apparent, but muscle weakness of the diaphram is the typical cause of mortality due to the increased inability to breath properly [1]. XLMTM falls under a category of diseases known as centronuclear myopathies. These are characterized by abnormally shaped muscle cells with centrally located nucleus as compared to normal muscle cells with polar located nuclei. XLMTM patients also show abnormal development of muscle and immature muscle cells [2].
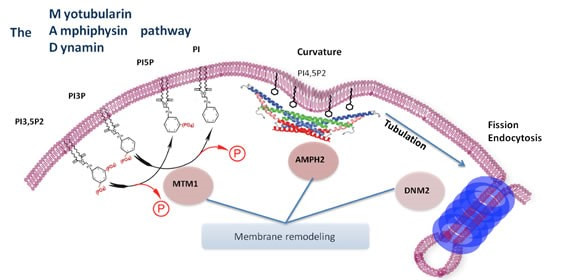
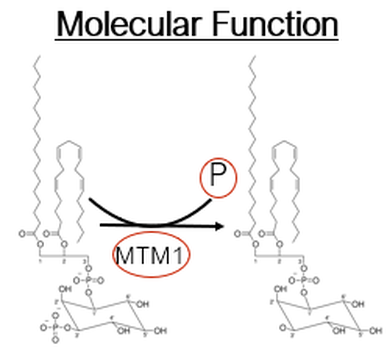
FunctionMyotubularin 1 is a protein tyrosine phosphatase involved in the dephosphorylation of phosphatidylinositol 3-phosphate (PI3P) and phosphatidylinositol 3,5-bisphosphate. (PI(3,5)P2) [4]. PI3P is a lipid signaling molecule located primarily around membranes, and in particular, membranes associated in an endocytic pathway [3]. The mechanistic relationship is not entirely understood but a negative feedback loop is proposed to regulate the formation and function of the endocytic vesicle. This relationship may be aberrant in myotubularin-1 defficient cells which is why muscle development is disregulated and muscle resembles fetal myofibers [5].
Fig 4. Myotubularin-1 role in endocytosis and membrane coupled processes
|
What is Myotubularin-1Myotubularin-1 is a protein tyrosine phosphatase involved in the dephosphorylation of lipid signalling molecules. In particular, Mtm1 acts on the membrane associated lipid signaling molecule phosphatidylinositol 3-phosphate (PI3P).
Fig 3. phosphatase function of myotubularin-1
|
Specific aims
Model organisms for studying myotubularin-1 mutations:
For these specific aims, the nematode worm C. elegans will be used, as it is an excellent model for studying muscle biology and has been highly studied in vesicle trafficking pathways and cellular fusion mechanisms. Two phenotypes will be assessed in these aims; those are vesicle trafficking shown in green fluorescence, and cell fusion shown in purple fluorescence. This model will be used to better understand the relationship between myotubularin-1 function and its role in trafficking and fusion.
For these specific aims, the nematode worm C. elegans will be used, as it is an excellent model for studying muscle biology and has been highly studied in vesicle trafficking pathways and cellular fusion mechanisms. Two phenotypes will be assessed in these aims; those are vesicle trafficking shown in green fluorescence, and cell fusion shown in purple fluorescence. This model will be used to better understand the relationship between myotubularin-1 function and its role in trafficking and fusion.
Aim 1
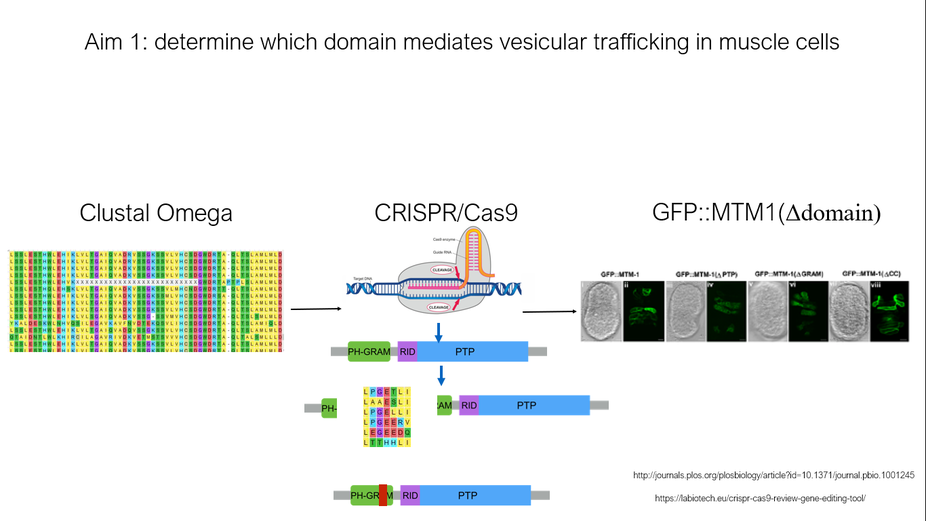
The goal of the first aim is to further characterize the mtm gene and to determine conserved amino acids in MTM1 that mediate vesicular trafficking in muscle cells. To identify these conserved amino acids, SMART and ClustalOmega will be used, Once conserved domain sites are identified, they will be knocked out via CRISPR/Cas9 in C. elegans and these mutants may then be screened for the inability to clear cell corpses. Those mutants that lack the ability to clear cell corpses, and thus cell fusion, will be screened for their ability to vesicle traffic. Vesicles expressing a GFP fusion will show localization. I hypothesize that of the various domains withing the gene, the ph-GRAM domain mutant may be important for mediating muscle cell fusion and vesicle trafficking because GRAM domains are common motifs in proteins associated with membrane coupled processes and signal transduction.
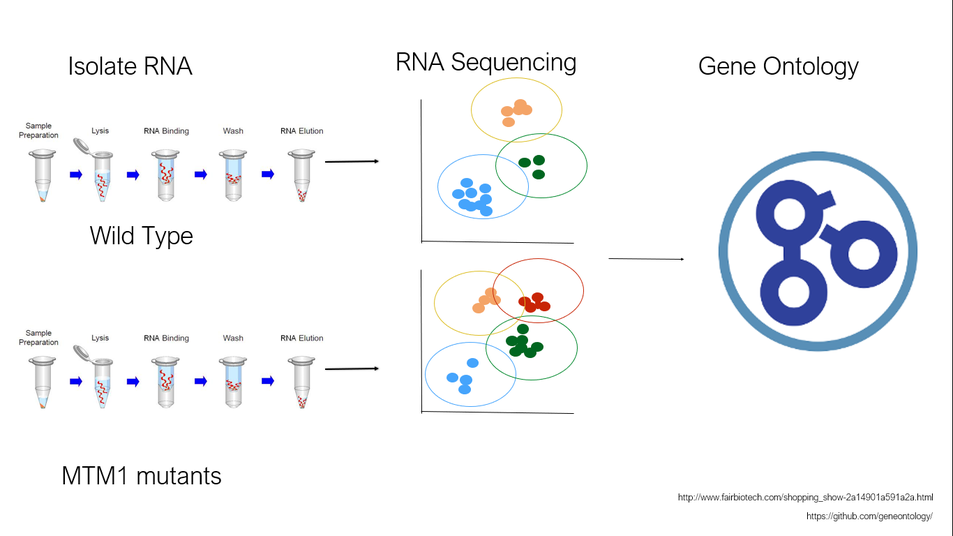
Fig 5. Basic workflow of aim 1
Aim 2
The purpose of aim 2 is to characterize the role of MTM1 in muscle cell fusion events. Again, using C. elegans as a model, RNA will be isolated from both wild type mtm1 expressing C. elegans and our mutated mtm1 knockout C. elegans from aim 1. This will be subject to RNA-seq for comparison. Using GO analysis to identify vesicle trafficking proteins that vary between WT and mutant organisms, will be subject to further mutagenesis. Their role in fusion will be screened for and observed by studying fusion defects in C. elegans. Genes that are found to be dysregulated will provide insight into how MTM1 mediates vesicle trafficking and fusion during development. Subsequent gene mutations will give insight into where myotubularin-1 lies in the fusion pathway. I believe, since the ph-GRAM domain is evolutionarily conserved in muscled organisms, this domain will be important in fusion. In GRAM mutants, proteins involved in trafficking and fusion will by dysregulated.
Fig 6. Identification of dysregulated proteins among mtm1 domain mutants
Aim 3
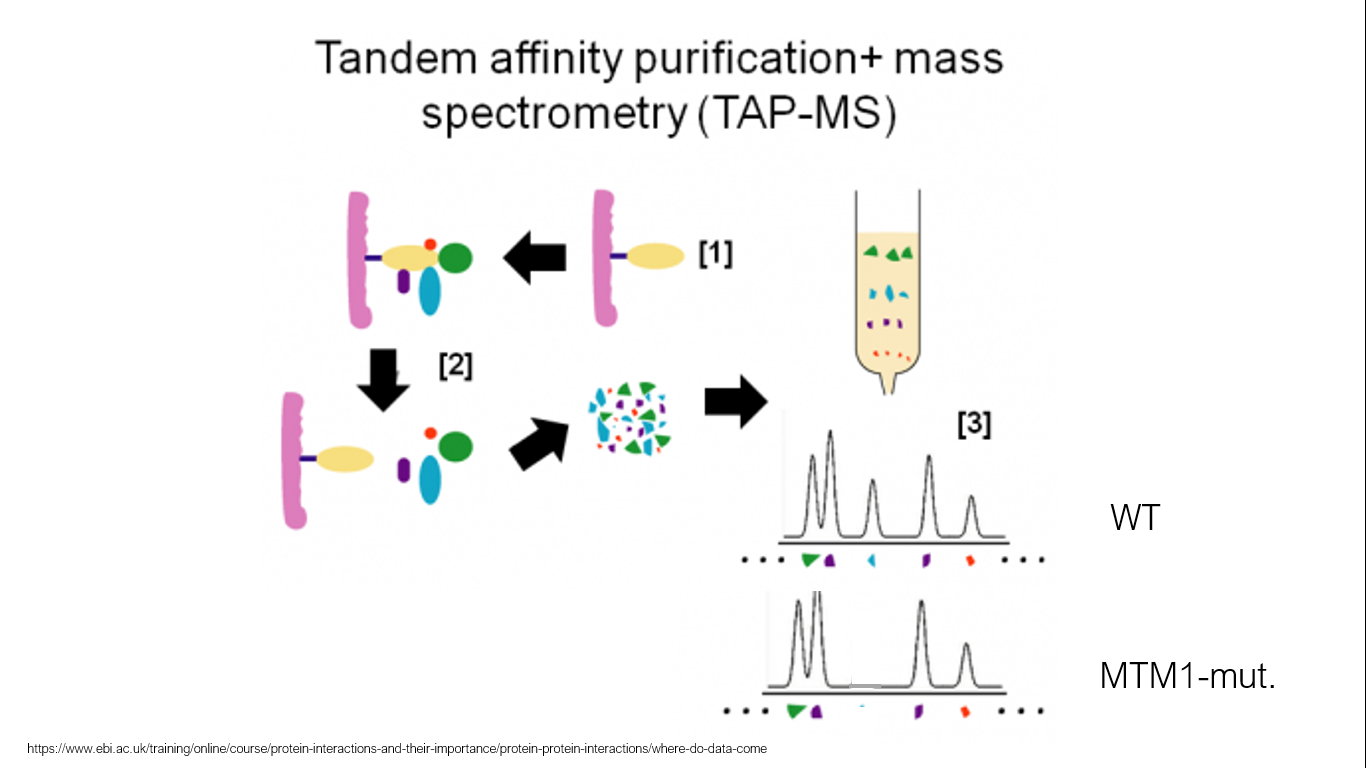
Aim 3 is designed to identify novel MTM1 muscle-specific fusion protein interactors. Different protein interactions will be analyzed by using tandem affinity purification with mass spectroscopy to identify protein interaction complexes. TAP-MS will be done on whole worms of WT and MTM1 mutants. Altered interacting partners with MTM1 between WT and MTM1-mutants will be subject to GO analysis to sort function. These found interacting partners will be used in an immunoprecipitation to better understand interactions at physiological conditions using a mouse model to better understand a mammalian system. Proteins identified will then be knocked out and muscle specific interctors will be able to be identified. Since myotubularin-1 is lowly expressed in healthy muscle, identifying novel protein interactors will elucidate why mtm1 mutations affect muscle so severely.
Conclusion and future directions
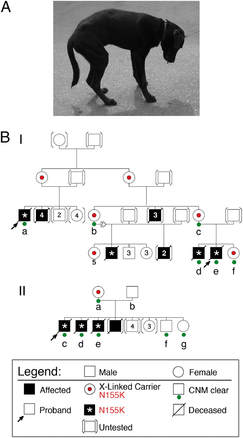
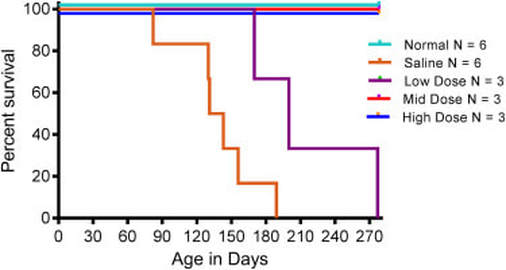
X-Linked myotubular myopathy is an incredibly rare, and currently incurable, congenital muscle disease resulting in premature death. More research into this gene and its functions are needed to fully understand the biology of this disease. Great advancements have been made in veterinary medicine however. Dogs and cats are also affected by this disease with equally severe consequences. Veterinarians have been developing gene therapy to replace the mutated gene with a functional copy. So far, the outcomes have been very positive resulting in whole body correction [6].
Final Draft
| final_presentation.pdf | |
| File Size: | 7056 kb |
| File Type: | |
Draft 1
| embj-35-561.pdf.crdownload | |
| File Size: | 877 kb |
| File Type: | crdownload |
References:
[1] https://ghr.nlm.nih.gov
[2] François Blondeau, et al; Myotubularin, a phosphatase deficient in myotubular myopathy, acts on phosphatidylinositol 3-kinase and
phosphatidylinositol 3-phosphate pathway, Human Molecular Genetics, Volume 9, Issue 15, 22 September 2000, Pages 2223–2229
[3] Gillooly, D J, et al. “Cellular Functions of Phosphatidylinositol 3-Phosphate and FYVE Domain Proteins.” Biochemical Journal 355.Pt 2 (2001): 249–258.
[4] https://www.uniprot.org
[5] Alan H. Beggs et al; MTM1 mutation associated with X-linked myotubular myopathy in Labrador Retrievers, Proceedings of the National Academy of Sciences Aug 2010, 107 (33) 14697-14702
[1] https://ghr.nlm.nih.gov
[2] François Blondeau, et al; Myotubularin, a phosphatase deficient in myotubular myopathy, acts on phosphatidylinositol 3-kinase and
phosphatidylinositol 3-phosphate pathway, Human Molecular Genetics, Volume 9, Issue 15, 22 September 2000, Pages 2223–2229
[3] Gillooly, D J, et al. “Cellular Functions of Phosphatidylinositol 3-Phosphate and FYVE Domain Proteins.” Biochemical Journal 355.Pt 2 (2001): 249–258.
[4] https://www.uniprot.org
[5] Alan H. Beggs et al; MTM1 mutation associated with X-linked myotubular myopathy in Labrador Retrievers, Proceedings of the National Academy of Sciences Aug 2010, 107 (33) 14697-14702